Abstract
Introduction: Increased toxicity with pegaspargase (PEG) in older and higher body mass index (BMI) patients (pts) with acute lymphoblastic leukemia (ALL) has recently led to dose capping practices. We assessed the influence of age, body surface area (BSA), and BMI on PEG-related toxicity and pharmacokinetics from two consecutive DFCI ALL Consortium trials without dose capping.
Methods: Patient (pts) aged 1 to <19 years (DFCI 05-001) or 1 to <22 years (DFCI 11-001) with newly diagnosed ALL were eligible for enrollment. Those who received PEG (2500 IU/m 2) were included in this analysis. Pts received 1 dose of IV PEG on day 7 of Induction and every 2 weeks for 15 doses post-induction. Serum asparaginase activity (SAA), considered therapeutic at >0.1 IU/mL, was assessed 4, 11, 18, and 25 days after the Induction dose and nadir SAA was assessed before each Post-Induction dose.
Asparaginase-related toxicities were prospectively assessed and graded by CTCAE version 3.0 (DFCI 05-001) or 4.0 (DFCI 11-001). Asparaginase toxicity for this analysis was defined as ≥1 of the following: pancreatitis, thrombosis, ≥grade 4 hyperbilirubinemia, ≥grade 4 hypertriglyceridemia. Allergy was analyzed separately (due to presumed dose independence). Height and weight at diagnosis were used for analyses. BMI categories were assigned using standard percentile ranges based on gender specific 2000 CDC growth charts. BSA was calculated using the Mosteller formula.
Univariate analyses evaluated the relationship of age, BMI, and BSA with asparaginase toxicity. Comparisons of toxicity across BMI and BSA categories were performed using a Jonckheere-Terpstra test. Categorical comparisons for dichotomized BMI and BSA utilized a Fisher's exact test or chi square test. The relationships between BMI and BSA with toxicity were explored using multivariable models.
Results: Between 4/2005-12/2011 802 pts enrolled on DFCI 05-001 and between 6/2012-6/2015 240 pts enrolled on DFCI 11-001. Both trials included random assignment of asparaginase formulation. In total 911 patients received pegaspargase during Induction and 351 during Post-Induction.
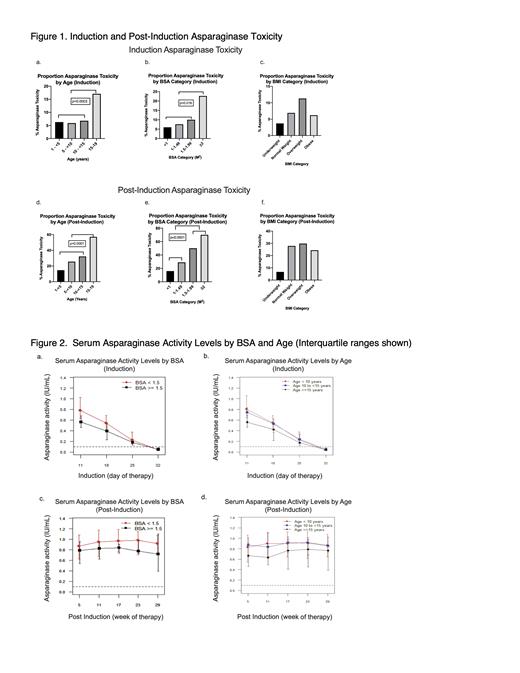
During Induction, pts ≥15 years of age had higher asparaginase toxicity rates (17.1% vs 6.2%, p=0.0003) (Figure 1a). Toxicity differed significantly across BSA categories (<1.5 m 2, 1.5 to <2.0 m 2, ≥2.0 m 2, p= 0.007) with increased toxicity in those with BSA ≥2.0m 2 (22.7% vs. 6.8% for those <2.0 m 2, p = 0.016) (Figure 1b). Age was highly correlated with BSA (Pearson r = 0.93, p <0.0001). There was numerically higher toxicity in the BMI category of overweight vs. those underweight or normal weight (11.3% vs 6.5%) however this did not extend to the obese category, and overall, increasing BMI was not associated with statistically higher toxicity (p= 0.13, Figure 1c).
Post-Induction, age ≥15 years was associated with increased asparaginase toxicity (57.1% vs 21%, p<0.0001) (Figure 1d). Toxicity differed significantly across BSA categories (p<0.0001) but was similar between BMI categories (p=0.19, Figure 1e-f). The impact of BSA was observed when dichotomized at thresholds of 1.5m 2 (54% vs. 19%, p<0.0001) and 2.0m 2 (70% vs. 23%, p=0.003) (Figure 1e). Considering only those ≥10 years of age, trends for BSA/BMI and toxicity were similar. There was no significant association between BMI or BSA and allergy. In multivariable analysis, BSA was a significant predictor of Post-Induction toxicity (OR 4.21, p<0.0001). Age was significant in the univariate setting (OR 1.14, p<0.0001) however due to high correlation with BSA, was not included with BSA in the multivariable model.
Post-Induction, median nadir SAA levels were ≥0.1IU/mL for all BSA and age categories. Median SAA was similar or lower at all time-points for those ≥15 years of age compared with younger children. Median SAA for pts with BSA ≥1.5m 2 were similar or lower compared to those with BSA <1.5m 2 (Figure 2a-d).
Conclusion: Age ≥15 years and BSA ≥2m 2 were each associated with significantly increased asparaginase toxicity. Older patients and those with higher BSA had similar or lower median SAA levels at all time-points. These results suggest that the differential toxicity seen in older patients and those with higher BSA is not explained by these patients having higher SAA levels. Prospective exploration of interventions to decrease toxicity in older patients and those with high BSA are needed.
Neuberg: Madrigal Pharmaceuticals: Other: Stock ownership; Pharmacyclics: Research Funding. Silverman: Takeda, Servier, Syndax, Jazz Pharmaceuticals: Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal